HIV is an important cause of dilated cardiomyopathy (DCM). Estimated prevalence rates of between 3% and 33% have been reported among HIV-infected children. Just like other systemic conditions associated with HIV infection, cardiovascular manifestations of the disease have been altered by the introduction of ART.
DCM is characterised by dilatation and impaired contraction of one or both ventricles. Systolic function becomes impaired and may result in heart failure.
The possible mechanisms of cardiomyopathy in HIV infection include; myocardial damage by HIV itself, autoimmunity, secondary infection, drug toxicity, co-infection with cardiotropic viruses (CMV, EBV, Coxsackie virus), nutritional deficiencies (selenium, vitamin B12) and low levels of growth and thyroid hormones.
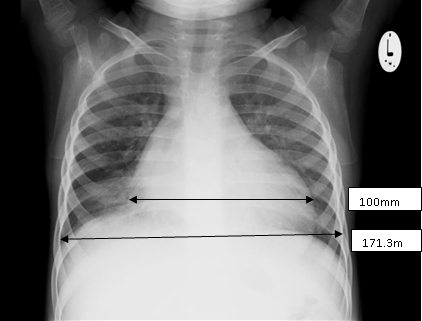
Diagnosis: The child may present with features of heart failure with evidence of cardiomegaly demonstrable on clinical examination, chest x-ray and electrocardiogram.
Echocardiographic findings include left ventricular (LV) dilatation, global hypokinesia and a reduced LV ejection fraction. Mitral and tricuspid regurgitation due to annular dilation may also be present.
Treatment: Antifailure medications (frusemide, spironolactone, captopril). To be managed in conjunction with the cardiologist. ART initiation is paramount.

HIVAN is defined by the presence of proteinuria associated with mesangial hyperplasia and/or global-focal segmental glomerulosclerosis, in combination with the microcystic transformation of renal tubules. Before the era of highly active antiretroviral therapy, more than 40% of HIV-infected children experienced renal complications. HIVAN is considered to be a renal disease induced directly by HIV-1. Black race is an established risk factor for the development of HIVAN.
Diagnosis: Patients with HIVAN typically present with significant proteinuria and rapidly progressive renal insufficiency in the setting of poorly controlled HIV infection marked by low CD4 counts and elevated HIV RNA levels. Most patients with HIVAN do not have significant oedema or hypertension. As proteinuria may be the first sign, it is advocated that all HIV-infected children should be screened for proteinuria at least once a year. Renal ultrasound may show large echogenic kidneys. However, in the case of long-standing kidney disease, there may be signs of fibrosis with small kidneys.
The definitive diagnosis of HIVAN requires a histological examination of renal tissues. In adults, HIVAN usually presents with the classic histologic findings of collapsing focal segmental glomerulosclerosis (FSGS) and tubular microcystic changes but in contrast, HIV-infected children more frequently show mesangial hyperplasia and/or classic FSGS in combination with the microcystic tubular lesions. Children with mesangial hyperplasia show a slower rate of progression of their renal disease when compared with children with classic or collapsing FSGS.
Treatment: ART is considered the best treatment for HIV. By reducing the viral load, ART may prevent progression of proteinuria and is associated with a marked improvement of HIVAN, resulting in slower progression to end stage renal disease (ESRD). HIV-infected children with HIVAN need to be managed in conjunction with a nephrologist. Diuretics, angiotensin-converting enzyme (ACE) inhibitors, and angiotensin-receptor blockers are other forms of supportive therapy available for HIV-related kidney disease in children but need to be used with caution.
In severe kidney damage, renal replacement therapy has been shown to offer improved survival. The most appropriate modality, peritoneal or haemodialysis depends on the availability of resources and expertise for the treatment. Necessary precautions need to be taken during dialysis to prevent the transmission of HIV-1 to health care workers.
HIV encephalopathy (HIVE) has been postulated to result from direct damage to the brain by HIV virus as it replicates in the CNS as well as viral/host interactions that lead to CNS damage by the release of soluble neurotoxic factors. In ART naïve children, prevalence of HIVE ranges from 20%- 60%.
Presentation: HIVE has a wide spectrum of manifestation and can be in form of progressive or static encephalopathy affecting motor, cognitive or language function. Motor involvement may include spasticity and movement disorder.
HIV associated progressive encephalopathy may be characterized by delay, loss or regression in developmental milestones and or neurological dysfunction. Children with static encephalopathy on the other hand have a non-progressive developmental delay and they may gain new skills but function below average.
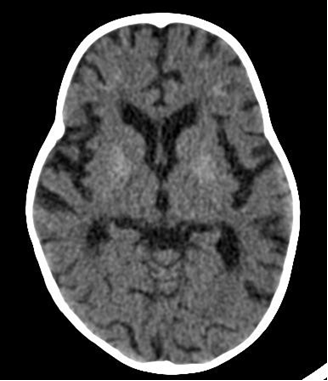
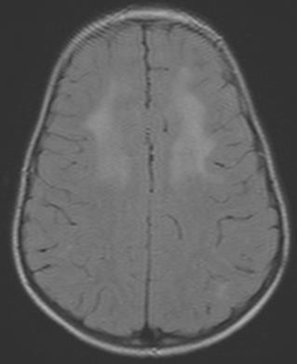
Diagnosis: Diagnosis is clinical, however, where available, neuro-imaging of the brain may show basal ganglia calcification, cerebral atrophy, and white matter changes.
Centers for Disease Control (CDC) case definition of HIV encephalopathy is: At least one of the following progressive features present for ≥2 months:
Treatment: Improvement occurs with institution of combination ART. However, ART may not fully reverse developmental dysfunction despite demonstration of clinical, immunological and virological response to therapy.