Pneumocystis pneumonia (PCP) is caused by a fungus called Pneumocystis jiroveci (formerly Pneumocystis carinii). PCP is the major cause of severe pneumonia (15–30%) and death (30–50%) in HIV-infected infants and sometimes in older children. The highest incidence of PCP in HIV-infected children is in the first year of life, with cases peaking at ages 3 to 6 months. ART and chemoprophylaxis with co-trimoxazole have led to about 80-90% decline in PCP (cases per 100 child-years). Young infants and severely immunocompromised patients are at high risk for PCP.
Clinical presentation
Pneumocystis jiroveci pneumonia may present as hypoxic pneumonia with cough, fever difficulty in breathing, tachypnoea and cyanosis. Onset can be abrupt or insidious with nonspecific symptoms such as poor feeding and weight loss. Some patients may not be febrile, but almost all will have tachypnoea by the time pneumonitis is evident on chest radiograph.
Differential diagnoses include: cytomegalovirus (CMV), other viral pneumonias, lymphoid intestinal pneumonitis (LIP), TB and Mycobacterium avium complex (MAC).
Diagnosis
Induced sputum, tracheal aspirate or bronchoalveolar lavage (BAL) are suitable samples for histology, direct immunofluorescence (IF) using monoclonal antibodies or PCR to detect Pneumocystis organisms. In one study, PCP was identified in 54% children using PCR, compared to 21% using IF and Grocott staining. Sputum and nasopharyngeal aspirates (NPA) are unsuitable for detection of PCP.
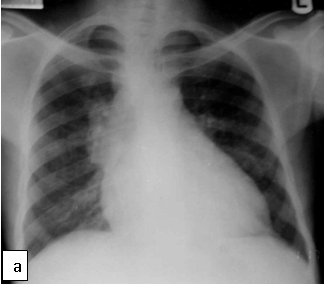
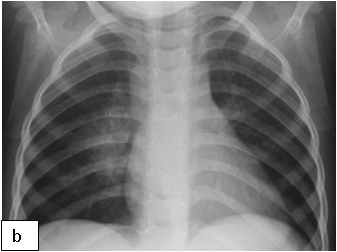
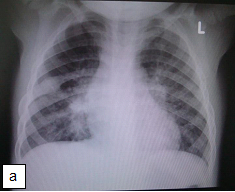
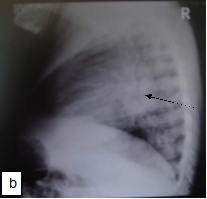
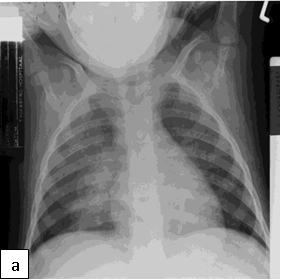
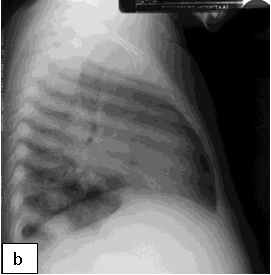
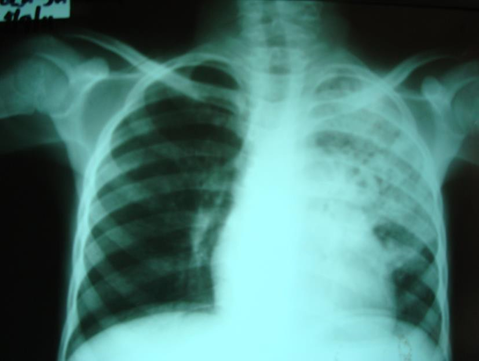
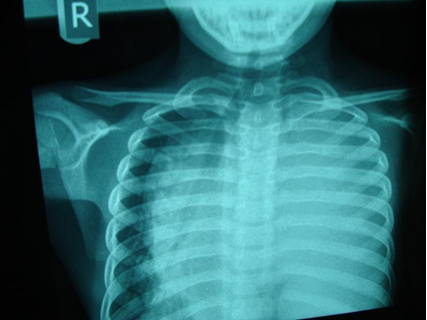
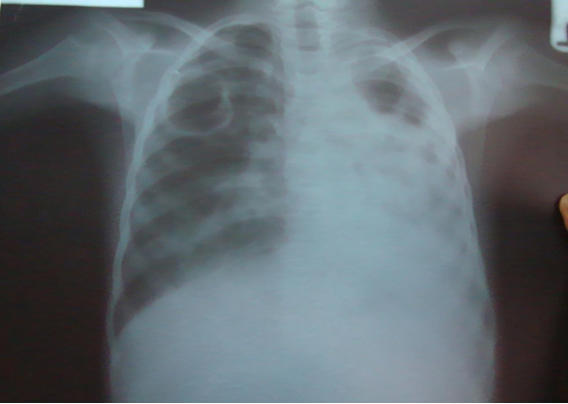
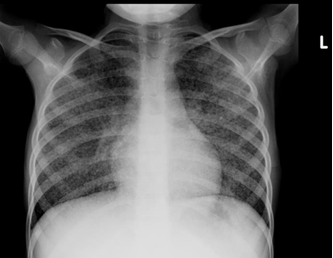
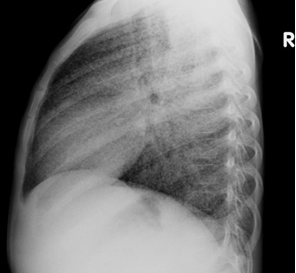
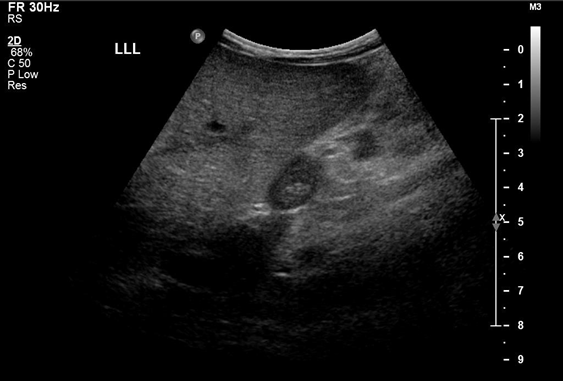
Chest x-ray is not usually diagnostic but may show bilateral diffuse parenchymal infiltrates with a “ground-glass” or reticulogranular appearance, but can be normal or have only mild parenchymal infiltrates. The earliest infiltrates are perihilar, progressing peripherally before reaching the apical portions of the lung.
Treatment
Empirical treatment with co-trimoxazole 15-20 mg/kg/d ÷ q6-q8 x 21 days is advocated in an HIV positive child with suspected PCP and treatment should not be delayed while awaiting results. The co-trimoxazole is given intravenously initially (where available) and stepped down to oral therapy when there is improvement. Supplemental oxygen is useful and a short course of corticosteroids is recommended in cases of severe PCP, starting within 72 hours of diagnosis. Prednisone is given at 1 mg/kg/dose twice daily for a week and tapered off over a week.
Alternative agents include pentamidine or clindamycin + primaquine. Intravenous pentamidine isethionate (4 mg/kg) once daily is recommended for patients who cannot tolerate co-trimoxazole or who demonstrate clinical treatment failure after 5 to 7 days of therapy.
PCP prophylaxis: co-trimoxazole administered (6 mg/kg) once daily. If syrup is unavailable, tablets may be used.
Co-trimoxazole is started from 6 weeks in HIV-exposed infants until HIV diagnosis is confirmed negative.
Co-trimoxazole is also indicated in the HIV-infected child with CD4 threshold severe for age and in any child who has had PCP.
Co-trimoxazole also protects against toxoplasmosis, malaria and serious bacterial infections in HIV-infected children.

There is increased risk of TB among HIV-infected children partly attributable to immunosuppression. M. tuberculosis is believed to enhance HIV replication; CD4 T-cells reduce with progressive HIV disease, vital for immunity to TB. There is also increased exposure to TB within among close family contacts.
HIV infection increases the risk of TB disease by a factor of 20. HIV-infected children are at increased risk of TB and of more severe forms compared with immunocompetent children and TB manifestations more severe in HIV-infected children, with reduced cure rates and increased mortality. In high HIV-prevalence areas, the recommendation is to test all children with TB for HIV and HIV counselling and testing is indicated in all TB patients.
Diagnosis
Cough, fever, weight loss are some of the clinical features of TB but are not specific for diagnosis of TB as these features may be seen in HIV infection. Disease progression may be more rapid and the development of complicated or disseminated disease is more likely in HIV-infected children. HIV-infected children may also have atypical findings, such as multi-lobar infiltrates and diffuse interstitial disease, and rapidly progressive disease, including meningitis.
Apart from TB, children with HIV infection may have other lung diseases related to their HIV infection. Bacterial causes include recurrent pneumonia; fungal – PCP; viral – CMV, adenovirus; other mycobacteria; non-infectious – lymphoid interstitial pneumonitis, bronchiectasis, pulmonary Kaposi sarcoma and cardiac causes - cardiomyopathy, pulmonary artery hypertension.
Tuberculin skin test (TST) or Mantoux test - may not be sensitive especially in severely immunosuppressed patients where there may be a false negative result.
Chest X-ray (CXR): TB enlarged perihilar lymph nodes are better visualised on a lateral CXR than a PA view, therefore, it is important to obtain both PA and lateral films when evaluating a child with suspected TB.
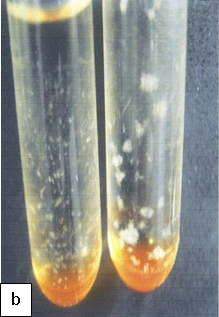
Bacteriologic confirmation: Sputum microscopy is positive in <10-15 % of children with probable TB. Yield from culture is <30-40%. Xpert MTB/RIF identifies twice as many TB cases as smear microscopy with a sensitivity of up to 79.4% and specificity of 96.5% against culture on one induced sputum. The Xpert MTB/RIF has facilitated rapid confirmation of childhood TB and diagnosis of drug resistant TB in Africa.
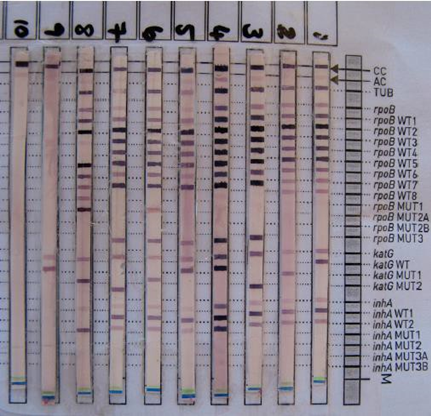
Xpert MTB/RIF (GeneXpert) is an automated nucleic acid amplification test that detects simultaneously TB and rifampicin resistance (a good and reliable proxy for MDR-TB) directly from sputum and other suitable fluids. GeneXpert test amplifies (by rapid, real-time PCR) and identifies targeted nucleic acid sequences in the TB genome in <2 hours and is more sensitive than smear (150 bacilli/ml cf 10,000 bacilli/ml). It is useful in the diagnosis of TB in HIV co-infected persons where the sensitivity of microscopy alone is low. Results from the Xpert MTB/RIF assay indicate whether or not MTB complex was detected in the sample. If MTB complex was detected, the results will also state whether resistance to RIF was detected, not detected, or indeterminate.
Line probe Assay (LPA), culture or Drug Sensitivity Testing (DST) is still required to confirm MDR-TB and perform other drug testing. LPA is a nucleic acid amplification test just like Xpert but is only performed on AFB smear positive and/or culture positive specimens. LPA can identify MTB and report on mutations that confer resistance to Rifampicin and INH.
Culture remains the gold standard for TB diagnosis. Culture is more sensitive than microscopy and Xpert, requiring a low organism load (10 bacilli/ml). Solid culture may require up to 6-8 weeks for incubation.


Treatment
Any child with active TB disease should begin TB treatment immediately (4 drug regimen – RHZE), and start ART as soon as tolerated in the first 8 weeks of TB therapy, irrespective of CD4 count and clinical stage.
Rifampicin can affect pharmacokinetics of some ARV medications especially non-nucleoside reverse transcriptase inhibitors (nevirapine) and protease 164 inhibitors (lopinavir). Co-administration may result in sub-therapeutic ARV drug levels. Options for antiretroviral therapy in TB co-infection include optimising the dose of nevirapine at 200 mg/m2, use of triple nucleoside reverse transcriptase inhibitor (AZT/3TC/ABC) or boosting with additional ritonavir for a lopinavir/ritonavir based regimen.
The bacillus Calmette-Guerin (BCG) vaccine contains a live attenuated strain of Mycobacterium bovis. There can be complications of immunization with bacillus Calmette-Guerin (BCG) in children in the setting of HIV infection. The World Health Organization in a revised consensus statement recommended that HIV infection in infants should not receive BCG vaccination. In practice, HIV DNA PCR testing is rarely performed during the first few weeks of life by which time immunization with BCG would have occurred.
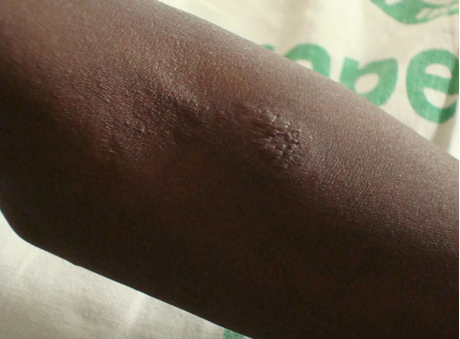
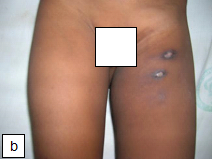
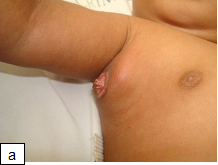
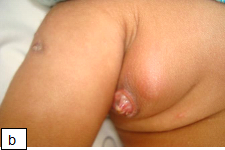
In one study, 6% of HIV-infected children who received intradermal BCG vaccination at birth developed clinically significant BCG complications after starting HAART, believed to be manifestations of the immune reconstitution inflammatory syndrome (IRIS). BCG complications occurring in HIV-infected children not receiving HAART usually involve localized disease manifesting as ulceration of the vaccine site with or without ipsilateral axillary lymphadenitis, and less frequently disseminated forms of disease in which M. bovis BCG is 176 confirmed in one or more anatomical sites far from both the site of injection and regional lymph nodes.
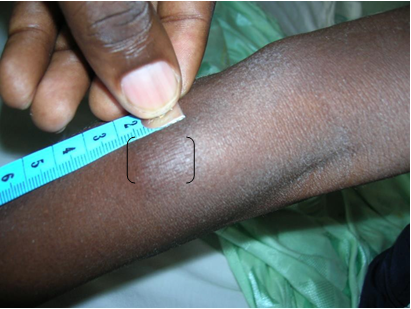
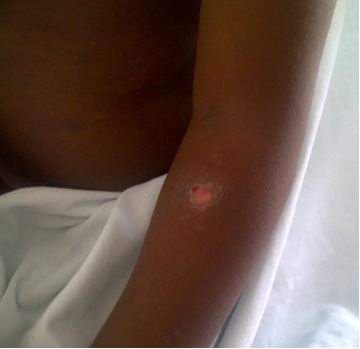
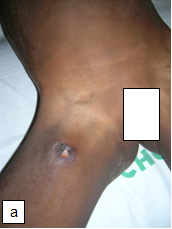
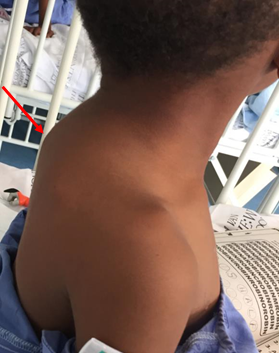
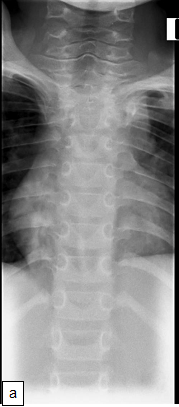
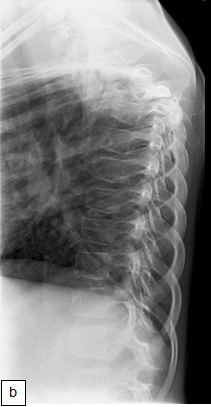
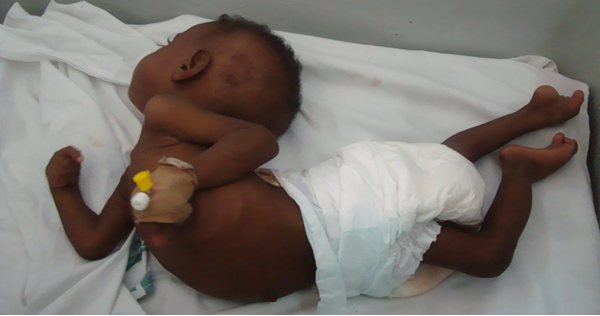
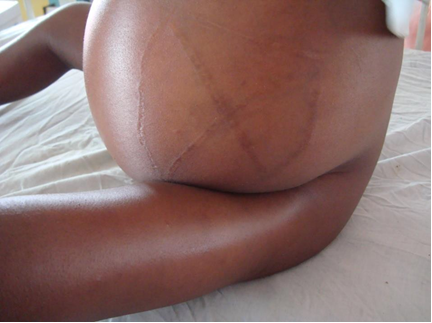
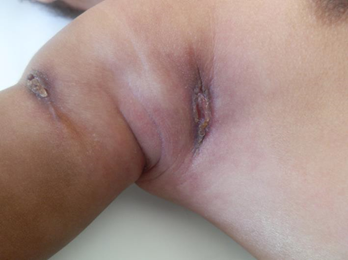
Diagnosis: The spectrum of presentations includes ulceration or abscess formation at the BCG vaccination site (right deltoid) and/or abnormally enlarged axillary lymph nodes with or without suppuration (regional disease). There may be spontaneous discharge of pus from the axillary abscesses. Abscesses in the ipsilateral supraclavicular and lower cervical regions (progressive regional disease) may also occur. Disseminated disease to other sites has also been reported.
Mycobacterial culture of material obtained from abscesses at the vaccination site, suppurative regional lymph nodes, or gastric lavage specimens may yield positive cultures for M. bovis. However, further identification of Mycobacterium bovis BCG by PCR is required. GeneXpert diagnoses mycobacterial species and is not specific to Mycobacterium tuberculosis (MTB). Therefore, if mycobacterial species are positive on GeneXpert, further identification should be requested if BCG disease is suspected.
Treatment: Spontaneous perforation and sinus formation usually occur if the abscess is left untreated. Needle aspiration helps to prevent these and shorten the duration of healing, apart from offering valuable diagnostic information. Sometimes repeated aspirations are required for optimal management, and wider-bored needles are preferred for ease of evacuation of thick inflammatory materials. Incision and drainage should be avoided as it increases the risk of sinus formation and delayed wound healing and unsatisfactory scar formation.
Antimycobacterial drugs for systemic disease:
Isoniazid (INH) 15 mg/kg/day
Mycobacterium avium complex (MAC) refers to multiple related species of non-tuberculous mycobacteria (NTM) (e.g., Mycobacterium avium, Mycobacterium intracellulare, and Mycobacterium paratuberculosis) that are widely distributed in the environment. MAC can appear as isolated lymphadenitis in both HIV-infected and HIV-uninfected children. Disseminated infection with MAC in paediatric HIV infection rarely occurs during the first year of life; its frequency increases with age and declining CD4 cell count, but can occur at higher CD4 counts in younger HIV-infected children than in older children or adults.
Clinical Manifestations: Respiratory symptoms are uncommon in HIV-infected children who have disseminated MAC, and isolated pulmonary disease is rare. Early symptoms can be minimal and may precede mycobacteraemia by several weeks.
Symptoms commonly associated with disseminated MAC infection in children include persistent or recurrent fever, weight loss or failure to gain weight, sweats, fatigue, persistent diarrhoea, and persistent or recurrent abdominal pain. Mesenteric adenitis may mimic acute appendicitis. Gastrointestinal symptoms can occur alone or in combination with systemic findings. Lymphadenopathy, hepatomegaly, and splenomegaly may occur.
Laboratory abnormalities include anaemia, leukopenia, and thrombocytopenia. Although serum chemistries are usually normal, some children may have elevated alkaline phosphatase or lactate dehydrogenase levels.
Diagnosis: Diagnosed by isolation of the organism from blood or from biopsy specimens from normally sterile sites (bone marrow, lymph node). Multiple mycobacterial blood cultures over time may be required to yield a positive result.
The volume of blood sent for culture also influences yield, with increased volume leading to increased yield. While histology demonstrating macrophage-containing acid-fast bacilli is strongly indicative of MAC infection, culture is essential to differentiate nontuberculous mycobacteria from M. tuberculosis, to determine which non-tuberculous mycobacterium is causing infection, and to perform drug-susceptibility testing. Testing of MAC isolates for susceptibility to clarithromycin or azithromycin is recommended.
Treatment: Combination therapy of MAC with a minimum of 2 drugs is recommended to prevent or delay the emergence of resistance, since monotherapy with a macrolide results in emergence of high-level drug resistance within weeks.
HAART should be initiated in children with MAC disease, considering the risk of IRIS. The drugs include either clarithromycin or azithromycin plus ethambutol; with clarithromycin as the preferred first agent, reserving azithromycin for patients with substantial intolerance to clarithromycin or when drug interactions with clarithromycin are a concern.
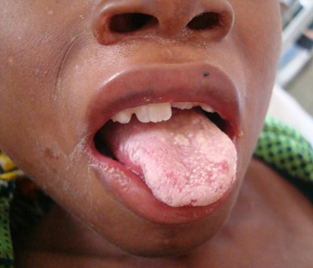
Candida spp causes the commonest fungal infections in HIV-infected children, which include oropharyngeal and oesophageal disease, vulvovaginitis, and diaper dermatitis. Once the organism penetrates the mucosal surface and widespread haematogenous dissemination occurs, invasive candidiasis ensues. This can result in candidaemia, meningitis, endocarditis, renal disease, endophthalmitis, and hepatosplenic disease. Oral thrush and diaper dermatitis occur in 50% to 85% of HIV-infected children.
Candida albicans is the most common cause of mucosal, oesophageal, and invasive candidiasis, but approximately 50% of reported cases of bloodstream infections in HIV-infected children are caused by non-albicans Candida spp., including Candida tropicalis, Candida pseudotropicalis, Candida parapsilosis, Candida glabrata, Candida krusei, and Candida dubliniensis. The non-albicans Candida species are important to recognize because several are resistant to fluconazole and other antifungals.
Clinical Manifestations: Thrush appears as creamy white, curd-like patches with inflamed underlying mucosa that is exposed after removal of the exudate. It can be found on the oropharyngeal mucosa, palate, and tonsils. Hyperplastic candidiasis comprises raised white plaques on the lower surface of the tongue, palate, and buccal mucosa and cannot be removed. Angular cheilitis occurs as red fissured lesions in the corners of the mouth.
Oesophageal candidiasis often presents with odynophagia, dysphagia, or retrosternal pain, and unlike adults, many children experience nausea and vomiting. Suspect oesophageal candidiasis when patients with oropharyngeal thrush have odynophagia or dysphagia. It may manifest in young children as drooling. Infants struggle to swallow when feeding.
Renal candidiasis presents with candiduria and ultrasonographically demonstrated renal parenchymal lesions, often without symptoms related to renal disease.
Diagnosis: Oral candidiasis can be diagnosed clinically as a whitish plaque on the buccal mucosa that comes off with difficulty on scraping. The yeast cells may also be seen with a potassium hydroxide preparation and culture with microscopic demonstration of budding yeast cells in wet mounts or biopsy specimens.
Oesophageal candidiasis has a classic cobblestoning appearance on barium swallow. Endoscopy is also helpful for ruling out other causes of refractory oesophagitis, such as HSV, CMV, and Mycobacterium avium complex. Candidaemia is best diagnosed with blood culture.
Treatment
Oropharyngeal candidiasis (OPC)
Early, uncomplicated infection can be effectively treated with topical therapy using clotrimazole troches or oral nystatin suspension 100 000u 4 to 6 hourly x 7 to 14 days. Oral therapy with fluconazole for 7 to 14 days is recommended for moderate to severe OPC. For fluconazole-refractory OPC, itraconazole oral solution should be used.
Oesophageal disease
Oral fluconazole 6 – 12 mg/kg/day x 14 to 21 days is effective for treatment of Candida oesophagitis. IV fluconazole, amphotericin B, or an echinocandin should be used for patients who cannot tolerate oral therapy. For fluconazole-refractory disease, itraconazole solution, voriconazole, amphotericin B, or an echinocandin are alternatives. Suppressive therapy with fluconazole three times weekly is recommended for recurrent infections.
Invasive disease
For invasive disease, investigation for a deep tissue focus of infection is important- in the form of echocardiogram, renal or abdominal ultrasound. Central venous catheters may need to be removed when feasible in HIV-infected children with candidaemia. The treatment of choice for invasive disease in HIV-infected children depends on severity of disease, previous azole exposure, and Candida isolate obtained (if known). Recommended duration of therapy for candidaemia is 14 days after documented clearance from the blood.

The prevalence of CMV infection among HIV-infected pregnant women is higher than in the general population and the HIV-infected infants have a 3-fold higher risk for symptomatic congenital CMV infection. CMV causes a wide range of diseases in HIV-infected children, including pneumonitis hepatitis, retinitis, encephalitis, oesophagitis and colitis.
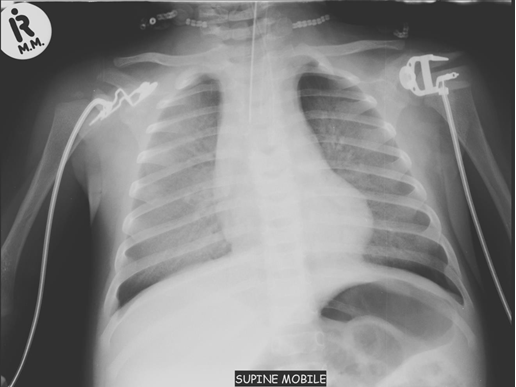
Presentation: The signs and symptoms of CMV disease often overlap with other infectious processes, therefore, the diagnosis of CMV disease in HIV-infected children should take into consideration clinical presentation and radiological finding along with laboratory testing. CMV pneumonia presents with fever, dyspnoea and hypoxemia. A chest radiograph shows diffuse pulmonary infiltrates (ground glass appearance) as seen in other viral pneumonias and PCP. The isolation of CMV from isolates including BAL does not prove that the child has CMV pneumonia. Co-infection with both PCP and CMV is common.
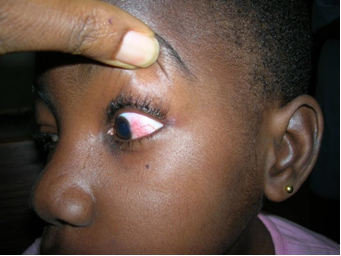
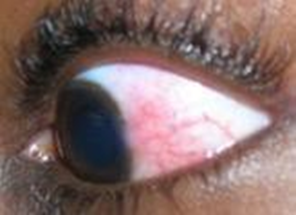
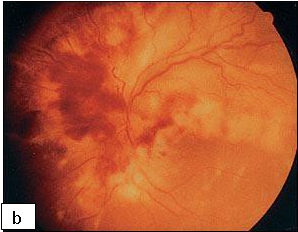
CMV produces a necrotic rapidly progressing retinitis with characteristic white perivascular infiltrate with haemorrhage (brushfire retinitis). Peripheral lesions may be asymptomatic, and even advanced disease does not cause pain. In children, strabismus or failure to fix and follow objects may be important clues to the diagnosis.
Diagnosis: Laboratory tests need to be interpreted in the clinical context as the virus, CMV DNA, and CMV antigen can all be detected in some patients who do not have active disease.
Quantitative PCR tests and the CMV pp65 antigenaemia test are available for detecting viral DNA and antigen, respectively. CMV viral load in blood is more useful for the diagnosis and monitoring of patients with CMV. There are no established cut-off values to definitively diagnose active CMV infection. Some experts base their decision to initiate antiviral therapy at viral load values of ≥4.0 log copies/ml in the presence of suggestive clinical features. In tissue invasive disease, the gold standard for diagnosing CMV disease is the identification of CMV inclusions or positive CMV-specific immuno-histochemistry staining on histopathology.
Treatment: Ganciclovir 5 mg/kg/dose IV 12 hourly, instituted in addition to ART. May switch to valacyclovir 15 mg/kg/dose 12 hourly when there is improvement and the patient can tolerate oral therapy. Total duration of therapy is 4 – 6 weeks.
An alternative drug for treating CMV disease or for use in ganciclovir-resistant CMV infections in HIV-infected children is foscarnet.
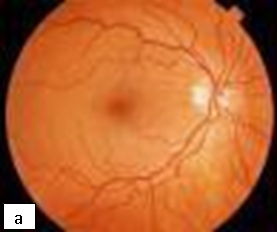
Cryptococcosis is fungal opportunistic infection caused by Cryptococcus neoformans which predominantly affects HIV-infected individuals with very severe immunosuppression. The use of potent antiretroviral therapies has resulted in a general decrease in the incidence of opportunistic infections associated with AIDS. Cryptococcosis is less frequent in children than in adults. Median age is about 10 years and median CD4/ cell count of <100 cells/μl at the time of diagnosis.
Clinical presentation: Most frequent – Meningoencephalitis typically presenting as a subacute process characterised by headache, fever, and later altered mental status as well as meningeal signs. Other manifestations include pneumonia and cutaneous diseases.
Laboratory evaluation: Culture of the organism is the gold standard for confirmation of the diagnosis of initial cryptococcal disease, confirmation of relapses or cases refractory to treatment, and adequate response to treatment.
Rapid diagnostic CrAg assays, either latex agglutination (LA) or lateral flow assay (LFA) to be used in cerebrospinal fluid (CSF), serum or plasma.
For cryptococcal meningitis prompt lumbar puncture (LP) with measurement of CSF opening pressure and rapid CSF CrAg assay is recommended or if access to CrAg assay is not available, CSF India ink test examination. In settings without immediate access to LP, or when it is clinically contraindicated, rapid serum or plasma CrAg is recommended.
Treatment
Meningeal and disseminated non-meningeal
Induction (x2 weeks) - Amphotericin B (0.7 – 1.0 mg/kg/dose/ daily IV) + flucytosine or Amphotericin B + fluconazole (12 mg/kg/day).
Alternatively, where amphotericin B is not available:
Consolidation (x 8 weeks): Fluconazole PO (6-12 mg/kg/day up to 400-800 mg/day if below 19 years).
Maintenance (secondary prophylaxis): Fluconazole PO (6 mg/kg/day up to 200 mg/day if < 19 years)
≤2 years of age: At least one year + evidence of immune reconstitution with optimal ART (CD4 >25% or absolute count >750 cells/μl).
<2 years of age: Anti-fungal maintenance treatment should NOT be discontinued.
Timing of ART initiation: To be deferred until there is evidence of a sustained clinical response to anti-fungal therapy, and after 2-4 weeks of induction and consolidation treatment with amphotericin B-containing regimens (+ flucytosine or fluconazole), or after 4-6 weeks of treatment with a high dose oral fluconazole induction and consolidation regimen.
Localized non-meningeal disease
12 mg/kg/day up to 800 mg/day if < 19 years) x 2 weeks, 6 mg/kg/day up to 400-800 mg/day if < 19 years) x 8 weeks, and continued maintenance with fluconazole 200 mg/day.
Progressive multifocal leukoencephalopathy (PML) is a demyelinating disease of the central nervous system that results from infection with JC virus (JCV), a neurotropic polyomavirus. Asymptomatic primary infection with JC virus occurs in childhood and antibodies can be found in 86% of adults. JCV remains latent in kidneys and lymphoid organs, but, in the context of profound immunosuppression as occurs in HIV infection, JCV can reactivate, spread to the brain, and induce a lytic infection of oligodendrocytes, the CNS myelin-producing cells. PML is relatively uncommon among children. Median age at HIV-associated PML diagnosis was 12 years in one review.
Clinical features: Paresis, speech abnormalities, gait disturbances, ataxia, cranial nerve palsies and seizures. CSF white cell count, protein and glucose are usually normal to slightly elevated in patients with PML.
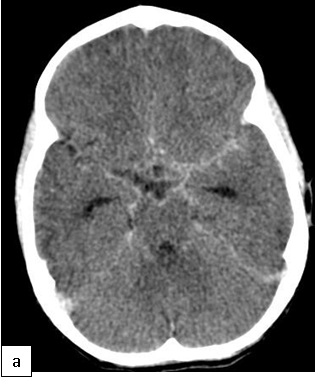
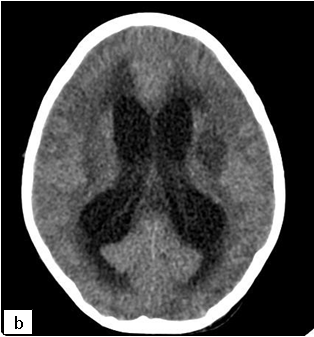
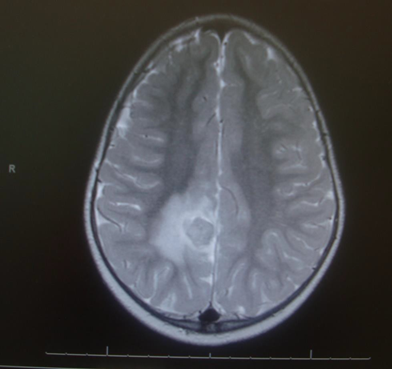
On cranial CT, PML lesions may appear as hypodense patchy or confluent white matter regions. Typical MRI features include single or multiple non-enhancing, non-space-occupying, predominantly white matter lesions commonly involving the frontal and parieto-occipital subcortical white matter.
Definitive diagnosis of PML can be established by detection of JCV DNA in the CSF or viral proteins on brain biopsy. Sensitivity of JCV DNA CSF PCR is as low as 59% but the specificity is about 100%.
Differential diagnosis: HIV encephalopathy, TB meningitis, tuberculoma, CMV infection, herpes simplex virus, cryptococcal meningitis and toxoplasmosis.
Treatment: The main stay of treatment is the initiation of effective ART which has been shown to improve survival in adults. PML-associated IRIS may occur which is a severe, often fatal, complication. Other antiviral agents have not shown any consistent benefits.