





Harry Teifel
ORCID ID: 0000-0002-6144-0275
Africa is set to become the most populous continent in the decades to come. In contrast to this – and with minor exceptions – only a small fraction of the Medical Devices deployed here are sourced from Africa. This anomaly is not sustainable in the medium term and begs several questions:
The purpose of this write-up is to offer a high-level glimpse into these three issues with a view to defining the requirements for sustainable and future-orientated Medical Devices industrialisation in Africa.
How does a country in Africa establish an eco-system, which supports the establishment of a vibrant and sustainable Medical Devices industry? Building on Michael Porter’s “Porter Diamond” (Porter, 1998), competitive advantage can be derived and improved by the optimal alignment of various aspects related to value creation, such as technology industry, skilled labour, and government support for a country’s economy.
Applying competitiveness enablement principles at a very practical level means dealing with two dimensions:
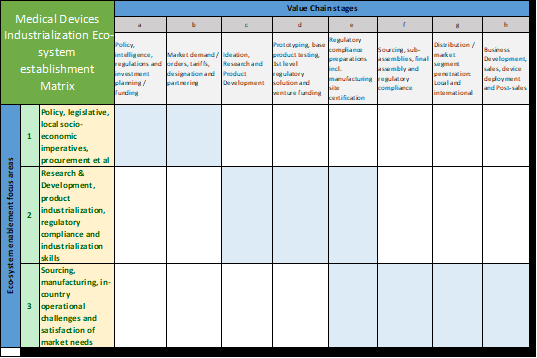
The proposed solution model sees the application of three primary industrialisation enablement focus areas overlayed with eight customized Medical Devices Value Chain stages:
In combination, this leads to the so-called “Medical Devices Industrialisation Ecosystem establishment Matrix”, Figure 1, where the shaded areas indicate which industrialisation enablement area applies to which stage in the Medical Devices Value Chain:
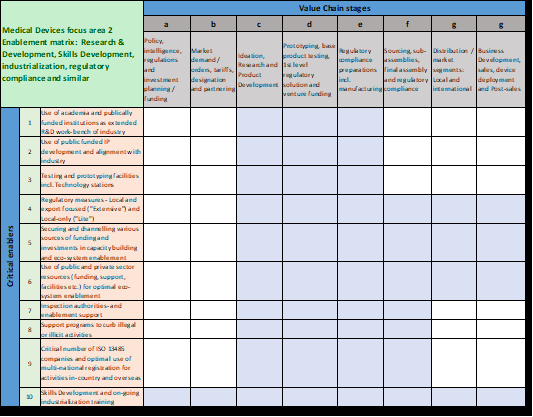
The next level of industrialisation eco-system development relates to the detailing of the three focus areas as outlined in section 2.1. For obvious reasons, the focus area to be detailed relates to “Research & Development, product industrialisation, regulatory compliance and industrialisation skills”. The following enablers are believed to be critical for enablement:
When the critical enablers of this focus area are aligned with a Value Chain perspective, this leads to the so-called “Medical Devices focus area 2 Enablement Matrix”, Figure 2, where the shaded areas indicate which industrialisation enabler applies to which stage in the Medical Devices Value Chain:
The establishment of a localisation-friendly Medical Devices eco-system is not a haphazard and laissez-faire occurrence, but instead requires multi-party buy-in, value chain thinking and the alignment of various measures for success.
One could be tempted to assume that industrialisation is largely a technocratic and mechanistic process, where an optimal “sausage machine” assures a constantly pipeline of successful Medical Devices. This is not the case! Instead, a variety of factors are at work and involving people – with their inherent strengths and weaknesses. Outcomes are much more difficult to predict, than one would imagine. Also, not all stages of the value chain are equally prone to failure with certain specific hand-over stages being particularly susceptible to ineffectiveness and problems. Welcome to the phenomenon called the “Valley of Death”.
There is an extensive body of knowledge dealing with the so-called “Valley of Death”, as the gap between invention and (implemented) innovation or the challenge of accelerating an innovation after proof of concept has been given.
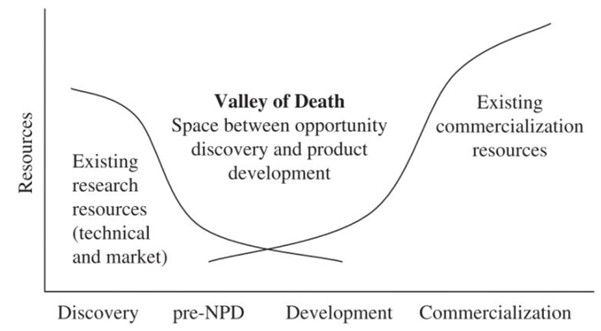
The Valley of Death, as described in Markham, Ward, Aiman-Smith, & Kingon (2010) is defined as the space between opportunity discovery and product development as shown in Figure 3. It occurs between the Product life-cycle stages of pre-New Product Development (pre-NPD) and completed Development:
Sandberg & Aarikka-Stenroos (2014) further assert that the Valley of Death phenomenon is most likely to occur in the following Technical Readiness Levels (TRLs):
This, in turn, leads to affected parties being confronted by the following type of challenges inhibiting successful industrialisation:
To illustrate the challenges of industrialisation, it is apt to create an analogy between Product Development and rugby with a multitude of "passes" between parties being necessary to achieve winning results. Simplistically put, the current game is characterized by undefined teams, poor passes and the ball often being dropped.
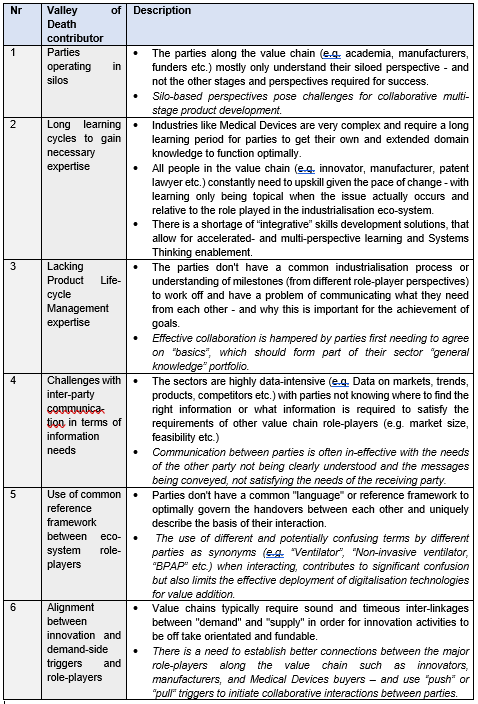
After extensive exposure to the phenomenon during Covid 19 and the personal involvement in a number of Medical Devices industrialisation projects, a list of the most prominent Valley of Death contributors has been created, Table 1:
Table 1: Most Prominent Valley of Death contributors
Equal emphasis should be given to both Valleys of Death, which are arguably equal in their negative effect on successful product industrialisation:
The following checklist, Figure 4, has been created to avoid the Valley of Death phenomenon from negatively affecting Product Industrialisation outcomes of Medical Devices:
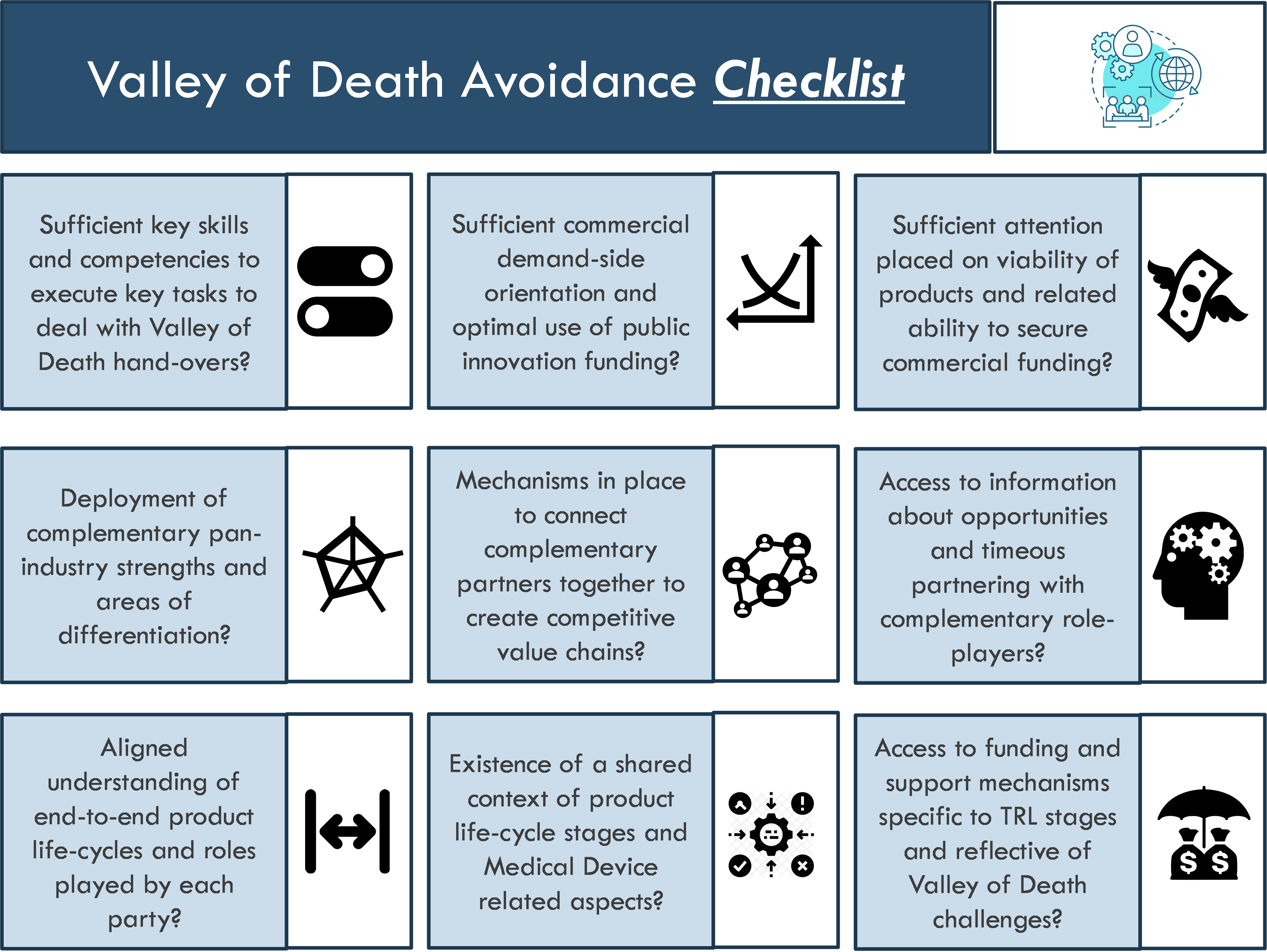
Similar to the establishment of localisation-friendly eco-systems being strategic and deliberate, so too is there a need for concerted Systems Thinking solutions to effectively counter-act and avoid the Valley of Death phenomenon from negatively inhibiting national and regional Medical Devices industrialisation.
Is it possible to deal with the significant challenges of industrialisation, as outlined in the previous chapters, without significantly altering the make-up of the industrialisation “system”? The author believes that product industrialisation value chains will increasingly experience seismic change through 4th Industrial Revolution (4IR) factors. This will necessitate a fundamental re-invention as to how parties collaborate to create value around new products. Current outcomes are simply not good enough – and “tweaking” existing industrialisation systems will not lead to the desired results.
As the world becomes increasingly globalized and competitive, people and organisations are challenged in the following areas, as shown in Figure 5:
In contrast to this, however, the there is a reduction in the available resources to deal with these increases, as shown in Figure 5, namely:
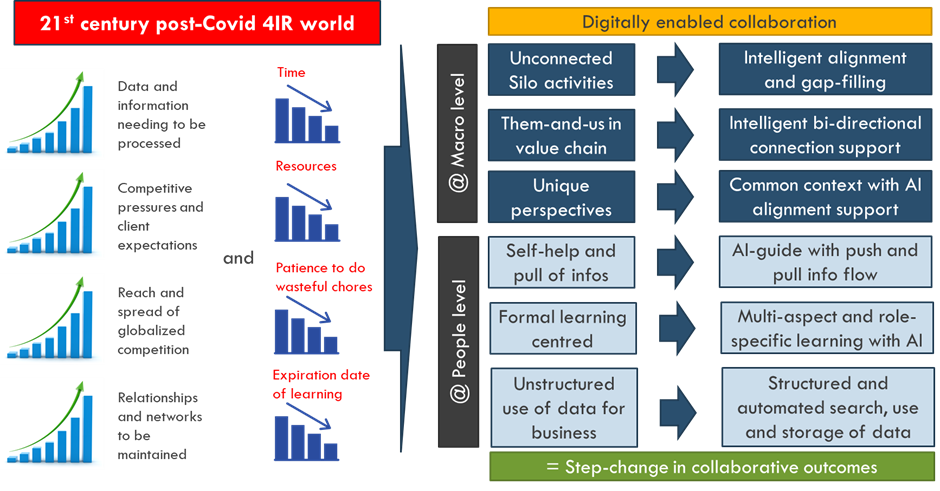
It is argued that the ever-increasing “imbalance” created will drive new forms of collaboration, both at a macro- as well as people level. This will herald a fundamental shift to digitally enabled collaboration, as the only way for the increased demands to be accommodated, as shown in Table 2:
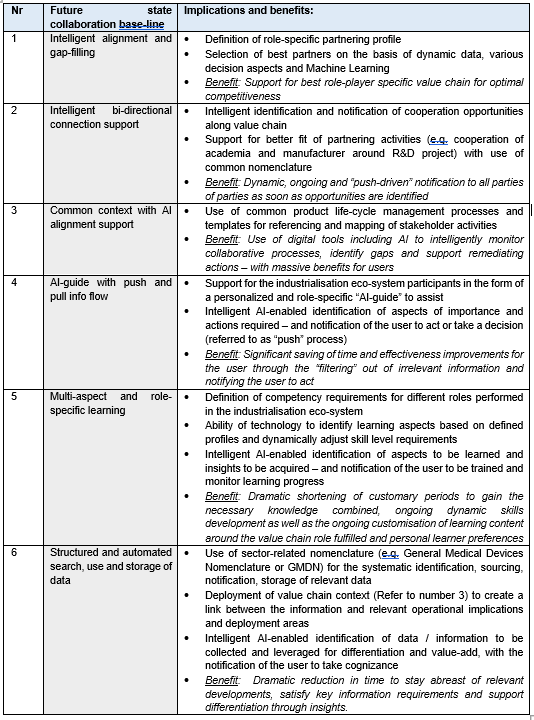
It is envisaged that the future state collaboration baseline, as described in the table above, will become an indispensable enabler to achieve the necessary level of competitiveness required for success in a 4IR environment.
Can future state industrialisation be achieved without technology? The author is of the belief that this is not possible, and that the improvement potential of existing multi-party industrialisation activities is limited.
Inherently, the limitation is human in nature and the ability of people to deal with the enormous and ever-growing challenges associated with industrialisation. Effectively the work requirements of people operating along the industrialisation value chain are so complex, specialized, time intensive and silo-specialized that a dramatic improvement is ONLY possible through the effective deployment of technology to support value creation of the individual parties – as well as their collaborative efforts.
People need support from technology for industrialisation outcomes to be dramatically improved!
It is not possible to train every party along the value chain up to be a Systems Engineer, who understands all interactions. Neither is it possible nor feasible to try and upskill an innovator to also be an expert on patent law – and vice versa.
It is argued that substantial improvements to industrialisation outcomes are only possible through the application of digital platforms or eco-systems, which allow parties to work together in a cloud environment and where is technology is systematically deployed to support people. The envisaged platform solution:
Industrialisation in the future is likely to be very different to today’s relatively ineffective and hit-and-mis practices. Continuous improvement of current industrialisation systems is unlikely to lead to breakthrough results. It is argued that dramatic improvements in industrialisation outcomes are only possible through the intelligent application of technology to deal with current systematic shortcomings and play a key enabling role in maximizing the ability of people to add value.
Africa needs to industrialize more Medical Devices and reduce its dependency on import. This process will not be easy – nor will it happen overnight. However, there are certain guidelines that the continent can draw from in charting its Medical Devices Industrialisation Roadmap:
It is hoped that the consideration of the pointers provided will in some way contribute to the development of a sustainable and future-orientated Medical Devices industrialisation in Africa.
Klitsie, J.B, Price, R.A., & de Lille, C. (2018). Overcoming the Valley of Death: A Design Innovation Perspective. In Next Wave: The 21st dmi: Academic Design Management Conference Proceedings (pp. 958-972). Design Management Institute.
Markham, S.K., Ward, S.J., Aiman-Smith, L. and Kingon, A.I. (2010). The Valley of Death as Context for Role Theory in Product Innovation. Journal of Product Innovation Management, 27: 402-417. DOI:10.1111/j.1540-5885.2010.00724.x
Porter, M. E. (1998). The competitive advantage of nations: With a new introduction. New York: Free Press.
Sandberg, B., & Aarikka-Stenroos, L. (2014). What makes it so difficult? A systematic review on barriers to radical innovation. Industrial Marketing Management, 43, 1293-1305.