





Lan Xu, Neil Stacey, David Rubin, Diane Hildebrandt
ORCID ID: 0000-0003-1342-4710, 0000-0003-4902-5201,
0000-0003-0316-9197, 0000-0001-7873-8855
School of Chemical Engineering
University of Witwatersrand
Gauteng
South Africa
Diabetes mellitus is one of the most common non-communicable diseases; approximately 0.72 million people in South Africa require insulin treatment. Insulin – a hormone peptide drug – is temperature sensitive and undergoes thermal fibrillation when exposed to temperature above manufacturers' recommendations. Most insulin formations must be stored at 2℃–8℃ in a refrigerator when unopened to maintain its potency and to keep insulin within manufacturers' suggested expiry date. However, one of the unforeseen consequences of frequent power outages in South Africa – and many other developing nations – is thermal denaturation of insulin within household refrigerators caused by frequent power outages. In addition, users cannot tell if insulin has reduced pharmacological action. This publication presents a preliminary design for an in-refrigerator medicine storage container utilising phase-change material to stabilise the temperature of insulin or other medications during power outages, and to provide a visual indicator if a melting event has occurred, even if refrigeration has subsequently been restored, to alert the user that their insulin may have expired. It is found that the principal variables of interest to determine the time to melting are the thickness of the layer of phase-change material in the unit's walls, and the degree of insulation of the outer shell surrounding the phase-change material. It is also determined that asymmetric insulation parameters for the inner and outer surfaces of the container's walls are a crucial design feature for usability, permitting an initial freezing time that is more rapid than the melting time. The current design has been found to be unable to meet all usability criteria, primarily due to an unacceptably long freezing time, regardless of the values of the available parameters. It is therefore concluded that design revisions are required to meet all usability criteria.
Keywords: Diabetes, medicine storage, phase-change material, public health
| Q | Rate of heat transfer |
| A | Area for heat transfer |
| ΔH | Change in enthalpy |
| M | Mass |
| t | Time |
| T | Temperature |
| U | Overall heat transfer coefficient |
| 𝑑 | Thickness |
| 𝑘 | Thermal conductivity |
| ℎ | Film heat transfer coefficient |
| 𝑑 | Thickness of material of construction |
| 𝛿 | Thickness of PCM layer |
The prevalence of diabetes in low- and middle-income countries has increased drastically over the past few decades – increasing urbanisation leads to physical inactivity and poor diet choices (Cullen, 2020). Within South Africa, 4 581 200 (12.8%) (International Diabetes Foundation, 2020) of the adult population has diabetes and about 15.5% of people with type 2 diabetes need insulin (Basu, et al., 2018). This issue is likely to worsen in the near future, as infection with COVID-19 has been found to be a risk factor for subsequent diabetes diagnosis (Barrett CE, 2022). Consequently, the number of new diabetes diagnoses is likely to increase above normal baseline for the duration of the pandemic. The public health needs of developing nations are not the focus of research and development (World Health Organisation, 2021), where there is a greater burden from increasing rate of diabetes cases and a lack of thermostable insulins. Moreover, limited access to reliable medication is exacerbated by insulin's cold chain requirements and frequent power outages.
Along the supply chain for insulin distribution, stakeholders such as manufacturers, distributors, and pharmacies have quality systems to maintain ideal storage conditions. Yet, when diabetics purchase insulin, the same standard is not maintained for several reasons (Braune, et al., 2018; Heinemann, et al., 2020). Currently, it is assumed that domestic refrigeration is reliable enough to store insulin, because developed nations have reliable power supplies. However, studies have shown that even during normal daily domestic refrigerator usage (without power outages) 11.3% of the time insulin degradation is an underestimated risk (Braune, et al., 2018). Insulin potency and quality is compromised by storing the insulin at nonideal temperatures. Most household refrigerators should operate between 0℃–8℃ for food safety, but may vary depending on position, air circulation, time, and age of the fridge. Therefore, temperatures within a household fridge are almost never ideal for medication storage, even when the mean temperature (6.1℃) is within insulin manufacturers' recommendation; the typical range (1.5℃–16.1℃ (James, et al., 2016)) is unsuitable.
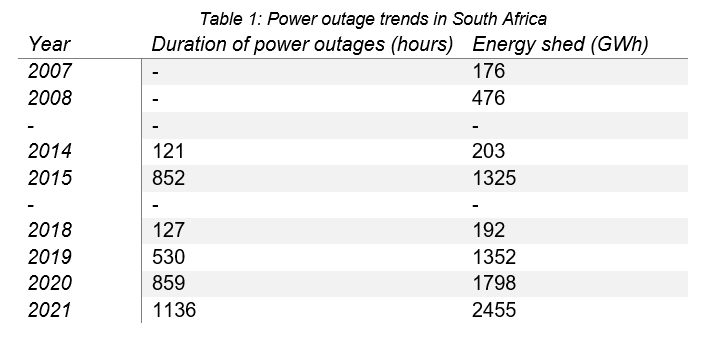
Coincidentally, many of the developing regions that experience frequent power outages have hot climates, compounding the issues caused by socioeconomic equality and access to reliable diabetes care. Regions with this burden in descending order are: Sub-Saharan Africa, South Asia, the Middle East, and some South American countries. In South Africa, frequent power outages, referred to as “load shedding”, are becoming more commonplace (Table 1) – a trend unlikely to change quickly. Therefore, with long and frequent power outages, it is likely diabetics will either waste money on more insulin to replace spoiled insulin, or accidentally use insulin with decreased potency – which is potentially life-threatening.
This compromised storage situation creates two urgent needs for insulin users in this context. The first of these is better thermal stability of storage and the second is a visual indicator when a given batch of medication has been exposed to out-ofrange temperatures, alerting a user to potential thermal degradation. This solution is also subject to several constraints imposed by the South African context. South Africa has a median per-capita household income in the region of R1 200 per month, severely constraining the acceptable cost of any accessories to treatment. There are also limitations on access to healthcare services, meaning that a solution must be lowmaintenance and not frequently require replacement parts. This means that common solutions to cold-chain management are unsuitable. Thermochromic stickers that detect out-of-temperature warming events are unsuitable because they would require replacement after each such event and would also require a cold chain for their distribution and delivery. Accurate digital temperature tracking, while potentially feasible, is comparatively costly and requires externalities including either battery replacement or charging, as well as either their own interface, or connectivity with outside electronic devices. Neither of these approaches to indicating out-oftemperature events address the core issue of thermal stability.
Our proposed solution is the use of a phase-change material (PCM) as a thermal stabiliser which, because of gravimetric and buoyancy properties, can also serve as a low-cost visual indicator by way of flotation of coloured beads of slightly differing density to the PCM. Such beads can be locked into one position base on the container's orientation during the freezing process and then, with the container's orientation altered for storage, only float to an alternate position if the phase change material melts. Hence, if there is a warming event there will be an irreversible positional change giving a visual indication. Afterward, re-freezing the material in the appropriate orientation will reset the beads' position for reuse.
The main usability criteria for this device are that it must have a sufficiently long time-to-melting, once solidified, to maintain thermal stability over the course of a sustained power outage. It must also have a sufficiently short time-to-freezing to allow users to freeze the phase-change material within a convenient amount of time. Ideally, the device should also hold enough excess thermal capacity to restore its internal temperature through multiple occasions of it being opened to retrieve insulin.
At this stage of the project, the objective is identifying the key design parameters and experimental targets for prototyping. This will be done on a theoretical basis by using a heat transfer model to identify which parameters affect the main usability criteria, and to determine whether the current design concept will be able to meet the criteria for usability, namely; a sufficiently long melting time to last through typical power outages, a sufficiently rapid freezing time for user convenience, and sufficient excess thermal capacity to repeatedly re-normalise temperature after the container has been opened to retrieve medication as needed.
The proposed phase-change material, C-14 paraffin, has a melting point of 6℃, latent heat of melting of 230kJ/kg, density of 763kg/m³, and thermal conductivity (in the liquid phase) of 0.14W/m*K. The proposed construction material for the device is polypropylene, which has thermal conductivity of 0.11 W/m*K. Air is assumed to have a heat capacity of 1.00kJ/kgK and a density of 1.2kg/m³. The convective heat transfer coefficient for air is taken to be 10W/m²*K.
The device is conceived to be a rectangular prism, with outer walls comprised of polypropylene, which will enclose a volume of C-14 paraffin as a phase-change material. For simplicity, the device can be envisioned as a cube, but the actual dimensions may vary. All six walls of the device are assumed to have the same dimensions and enclose an equal thickness of PCM. Final design may deviate from this approach, as it may turn out that different properties of convection from vertical and horizontal surfaces may require that the respective wall thicknesses of those surfaces differ.
For purposes of modelling, the following simplifying assumptions are made:
The device will be closed for storage, which means that heat transfer will occur through the outer skin only. Based on Fourier's Law, and the definition for an overall heat transfer coefficient (these are defined in equations 4 and 5), the rate of heat transfer during melting is given by:

During freezing, the device will be open, so the inner surfaces will also interact with the interior air of the refrigerator, so the rate of heat transfer is then given by:

Based on the principle of conservation of energy, the following energy balances can be formulated, governing freezing and melting:

Equation 3 relates the quantity of material and/or frozen to the enthalpy change that has occurred, while Equation 4 relates the enthalpy change to the heat transfer over time. Equation 2 relates the heat transfer rate to the temperature differences and hence, these equations together are sufficient to determine the rate of melting and/or freezing once the parameters are populated. The temperature parameters are a product of the outside conditions, while the other parameters are either material properties or are functions of the design geometry of the box.
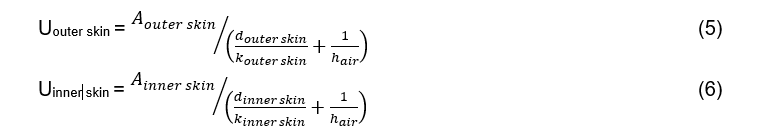
Conveniently, because the PCM is at a fixed temperature during freezing or melting, when a constant internal air temperature is assumed for the refrigerator, both processes take place at a constant rate, and the freezing and melting times can be estimated as follows:

Now, the total mass of the PCM is given by the total surface area of the container walls multiplied by the thickness, δ, of the PCM volume, multiplied by its density. Consequently, the above equations can be summarised as follows:
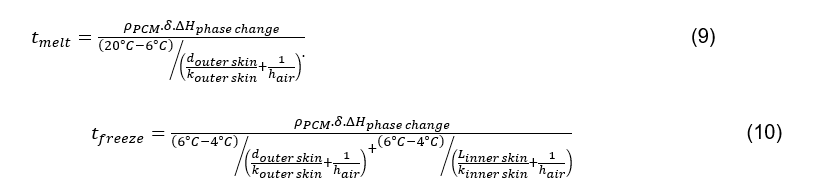
Equations 9 and 10 only have the thicknesses of the inner and outer skins, and the thickness of the PCM volume as independent variables, which makes it possible to assess two major usability criteria just in terms of those independent variables. This simplification is possible only if the inner and outer skin areas are close to equal, and only for simple geometries where outer surface area multiplied by thickness gives the total volume. For more complex geometries with heat transfer features to modify the respective surface areas, this simplification is not applicable.
The other critical usability criterion is tolerance to opening and closing of the unit for retrieval of medicine. There will be considerable user-variability in how much additional heat exchange takes place with each such event. However, it can be reasonably assumed that most users will open the container only briefly during a power outage, to conserve the cold environment inside. Therefore, the worst-case scenario among the likely use cases is that when the container is opened, all of its air contents are replaced with interior air from inside the refrigerator and then the container will be closed with air at the refrigerator temperature now inside. It can be fairly assumed that heat transfer between the storage container and now-enclosed air will be rapid enough to prevent medicines warming above the desirable range and that the temperature inside the container will re-stabilise at 6℃ in a reasonable timeframe. Hence, the area of concern is that the excess energy of the enclosed air will be added to the PCM each time the container is opened and closed.
This performance characteristic can be expressed as a fraction of the PCM volume that will be melted to re-stabilise temperature after a single opening event, and can be estimated using the following equation:
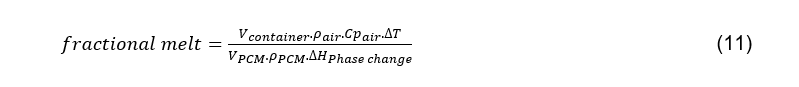
The only variables remaining in this equation are the volumes of the container interior and that of the PCM; the other parameters are physical constants. Hence, by substituting the known physical constants, the fraction of phase-change material required to melt to normalise temperature after an opening event can be expressed as the following linear relationship to the ratio of interior container volume to PCM volume:

The fractional melting per usage, as defined by Equation 12, is 0.0001.Vratio. For a container of square base measuring 18cm on each axis, a height of 12cm, PCM thickness of 1cm, the interior volume will be 3888 cubic cm, and the PCM volume will be 805 cubic cm, and thus the fractional melting per opening and closing event will be only 0.00048, or roughly 0.05%. This means that the overall excess thermal capacity available to compensate for opening/closing of the box will be far in excess of what is required.
This implies that the main consideration for opening/closing is in fact just ensuring that the interior skin's heat transfer is rapid enough that warmer interior air is cooled quickly enough to prevent much temperature rise in the medicines themselves. The low heat capacity of the air volume inside the container suggests that this will not be especially concerning and, therefore, if a user is careful to not keep the container open for extended periods during a power outage, the opening and closing of the container will not significantly affect its performance.
Because of this, heat incursion of this type is not a severe design constraint, leaving the volume of PCM relative to the interior volume of the container as a free variable, subject to other constraints.
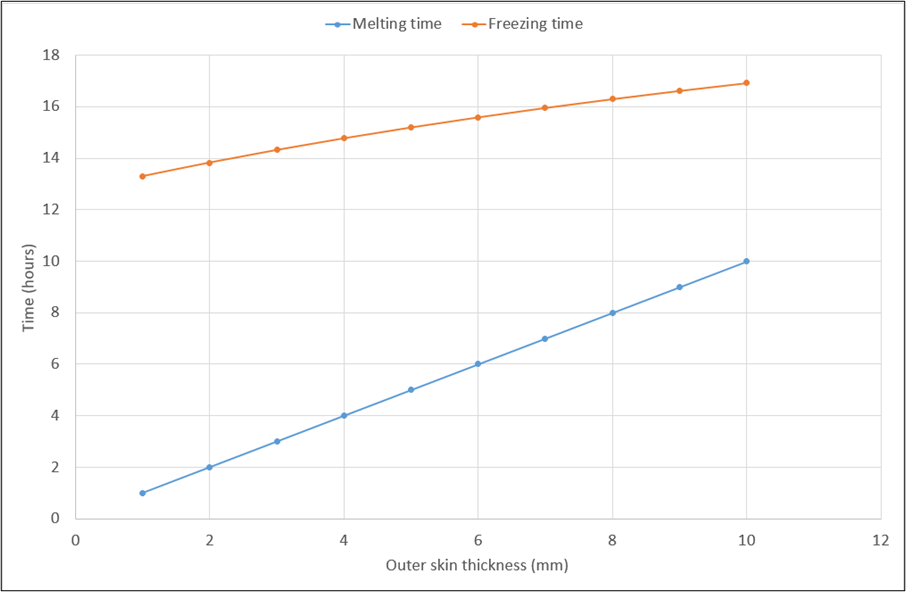
Figure 1 demonstrates quite clearly that the design, as currently conceptualised, is not able to meet all usability criteria. While a suitable melting time is achievable, albeit with a comparatively thick outer skin, the freezing time is unacceptably high. Freezing time is directly proportional to thickness of PCM volume but lowering freezing time by reducing 𝛿 will also commensurately reduce melting time, adversely affecting another key usability criterion.
Reducing inner skin thickness would also reduce freezing time, but not by a significant enough amount to yield a viable design, because the limitations of convective heat transfer will still apply and so, the heat transfer through the inner skin cannot be increased by that large a factor through that modification. Moreover, decreasing the inner skin thickness to less than the 1mm thickness currently envisioned risks compromising its structural strength. Increasing the outer skin thickness much beyond the 1cm thickness that is the maximum value in Figure 1 is also not viable because it will drive up the cost of the unit and make it excessively bulky.
The fundamental reason for the difficulty in achieving both of these usability criteria resides in the respective driving forces for heat transfer during melting and freezing. During freezing, the temperature difference between the refrigerator interior and phase-change material at its freezing point is very small, as low as 2℃, resulting in a very small driving force for removing heat from the PCM. During melting, the temperature difference, and therefore the resulting driving force, is considerably larger.
Consequently, fundamental design revisions are required to meet all usability criteria. Modifications that would potentially improve the relationship between the melting time and freezing time are as follows:
It has been shown that a storage container utilising C-14 paraffin as phasechange material is potentially feasible as a means of thermally stabilising medication stored in a household refrigerator during power disruptions, as a sufficiently long melting time is achievable with reasonable design parameters. However, it has been found that an unreasonably long freezing time affects the usability of the proposed design and, consequently, design revisions are needed in order to meet the required usability criteria. It has also been shown that the main parameters of interest for usability are the thickness of the PCM layer and the heat transfer coefficients for the inner and outer skins respectively. It has been found that the inner skin heat transfer coefficient must be considerably higher than that of the outer skin in order to meet the usability criterion of freezing time being shorter than melting time. Once a revised design that meets the usability criteria is developed, prototyping and laboratory testing should be focused on determining those heat transfer coefficients and achieving values for those parameters that meet the specified usability criteria.
The authors would like to acknowledge the funding provided by MerSETA for the MediVentors Consortium, and the contributions of Wits University.
Barrett CE, K. A. A. P. e. a., 2022. Risk for Newly Diagnosed Diabetes >30 Days After SARS-CoV-2 Infection Among Persons Aged <18 Years — United States, March 1, 2020–June 28, 2021. MMWR Morb Mortal Wkly Rep, Volume 71, p. 59–65.
Basu, S. et al., 2018. Estimation of global insulin use for type 2 diabetes, 2018–30: a microsimulation analysis. Lancet Diabetes Endocrino, 7(1), pp. 25-33.
Braune, K. et al., 2018. Storage Conditions of Insulin in Domestic Refrigerators and Carried by Patients—Insulin Is Often Stored Outside Recommended Temperature Range. American Diabetes Association, pp. 1-7.
Cullen, M., 2020. Type 2 Diabetes in Developing Countries. [Online] Available at: https://borgenproject.org/diabetes-developing-countries/
Heinemann, L. et al., 2020. Insulin Storage: A Critical Reappraisal. Journal of Diabetes Science and Technology, pp. 1-13.
International Diabetes Federation, 2021. IDF Diabetes Atlas: 10th Edition (South Africa Diabetes report 2000–2045). [Online] Available at: https://diabetesatlas.org/data/en/country/185/za.html
International Diabetes Foundation, 2020. IDF Africa Members. [Online] Available at: https://www.idf.org/our-network/regions-members/africa/members/25- south-africa [Accessed 2021 August 15].
James, C., Onarinde, B. A. & James, S. J., 2016. The use and performance of household refrigerators: a review. Comprehensive Reviews in Food Science and Food Safety, pp. 160-179.
World Health Organisation, 2021. Keeping the 100-year-old promise making insulin access universal, Geneva: World Health Organisation.